Neurodegenerative Diseases

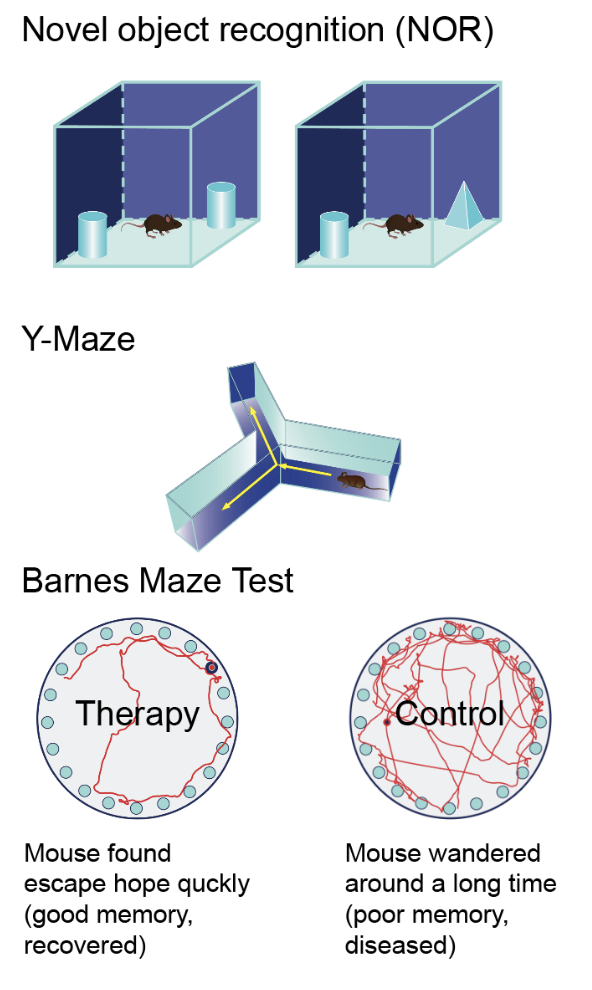 Novel Object Recognition (NOR) Test: This memory test has two parts. First, mice explore an object to learn it. Later, one object is replaced with a new one, and we record how long the mouse spends near each. We calculate a Recognition Index: (Time at new object – Time at old object) / Total time. This index ranges from -1 to 1. Higher values (more time at the new object) mean better memory. Lower values suggest memory issues, as mice naturally prefer to explore new things.
Novel Object Recognition (NOR) Test: This memory test has two parts. First, mice explore an object to learn it. Later, one object is replaced with a new one, and we record how long the mouse spends near each. We calculate a Recognition Index: (Time at new object – Time at old object) / Total time. This index ranges from -1 to 1. Higher values (more time at the new object) mean better memory. Lower values suggest memory issues, as mice naturally prefer to explore new things.